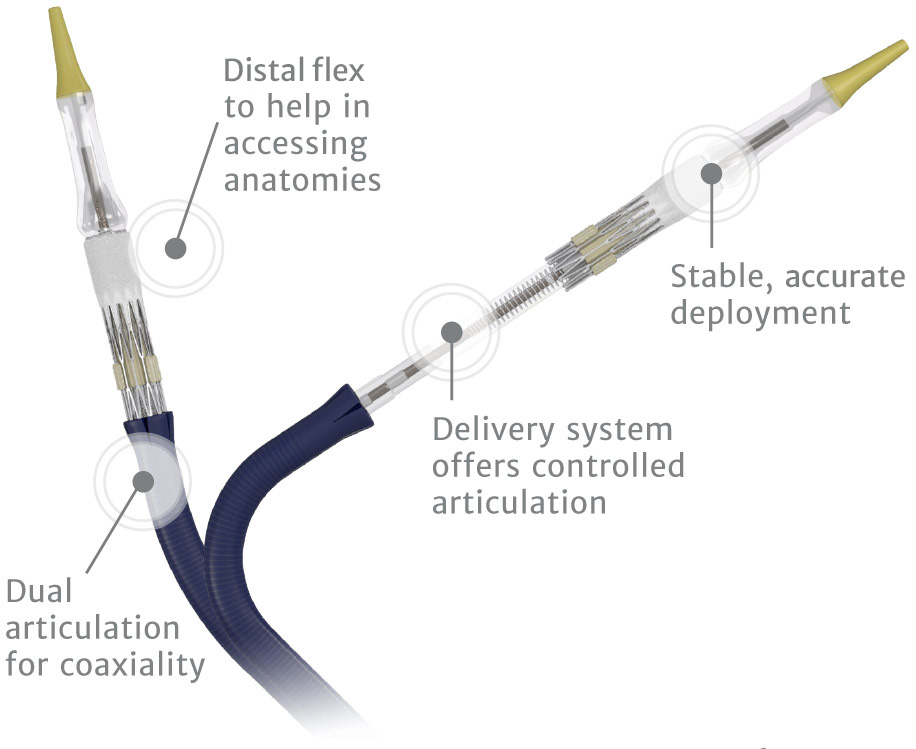
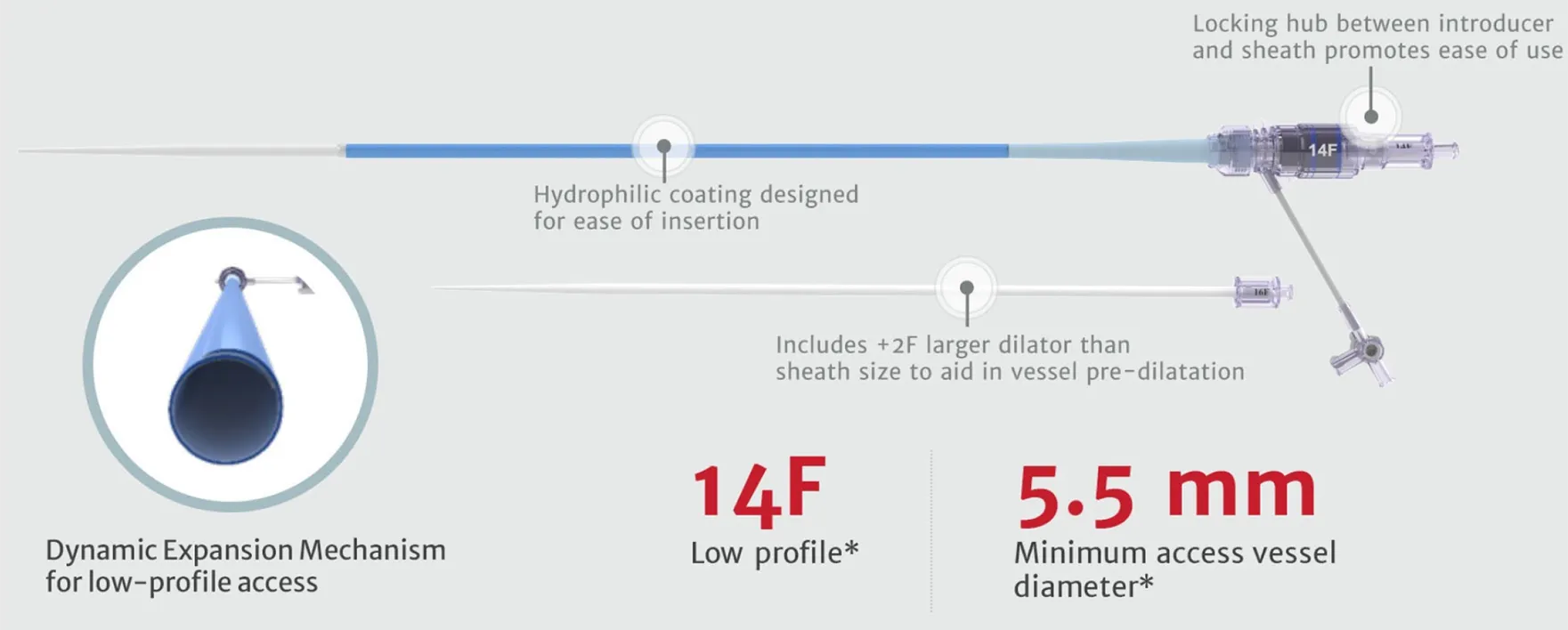
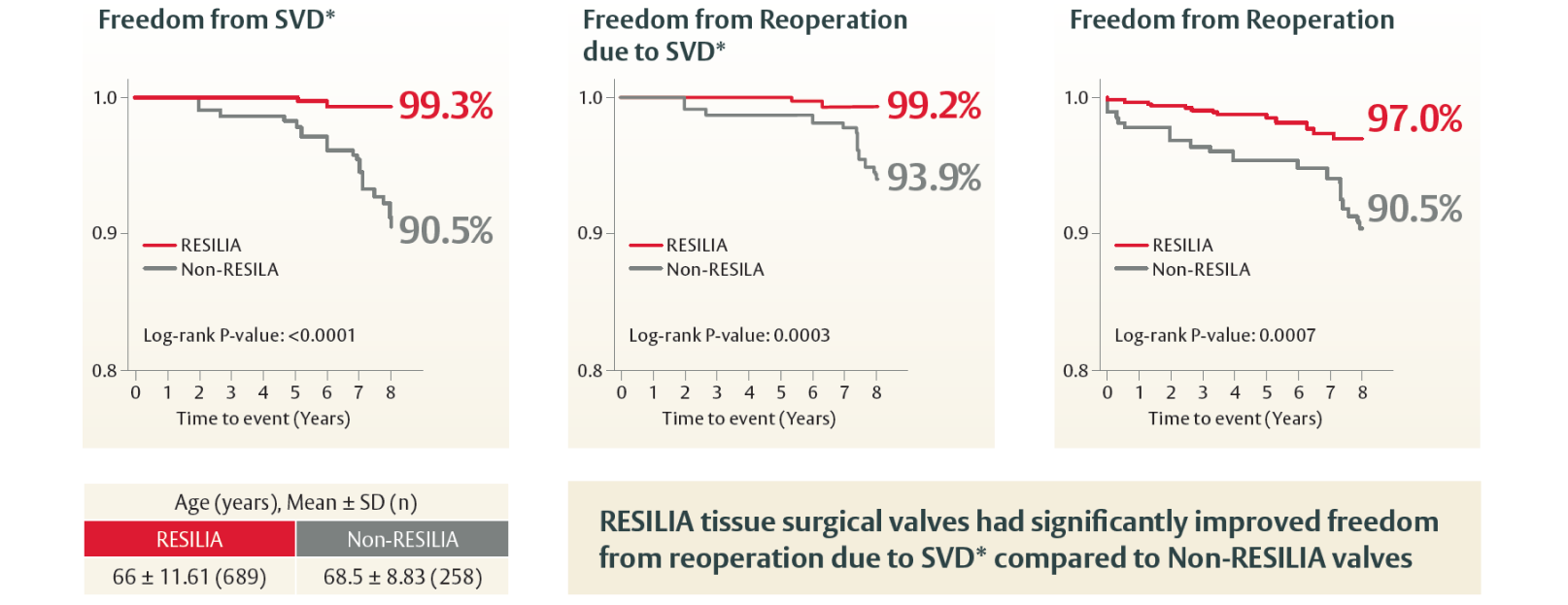
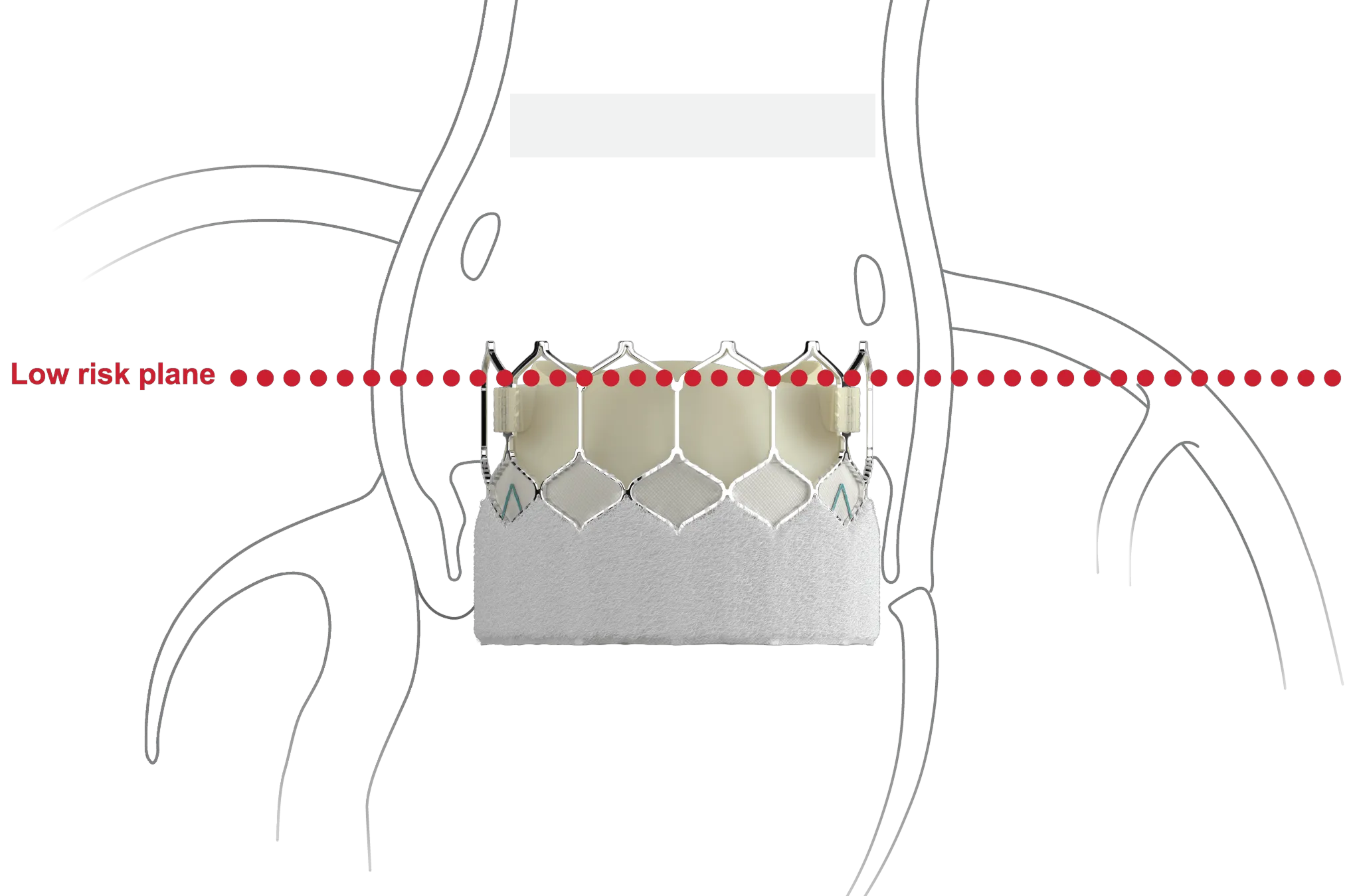
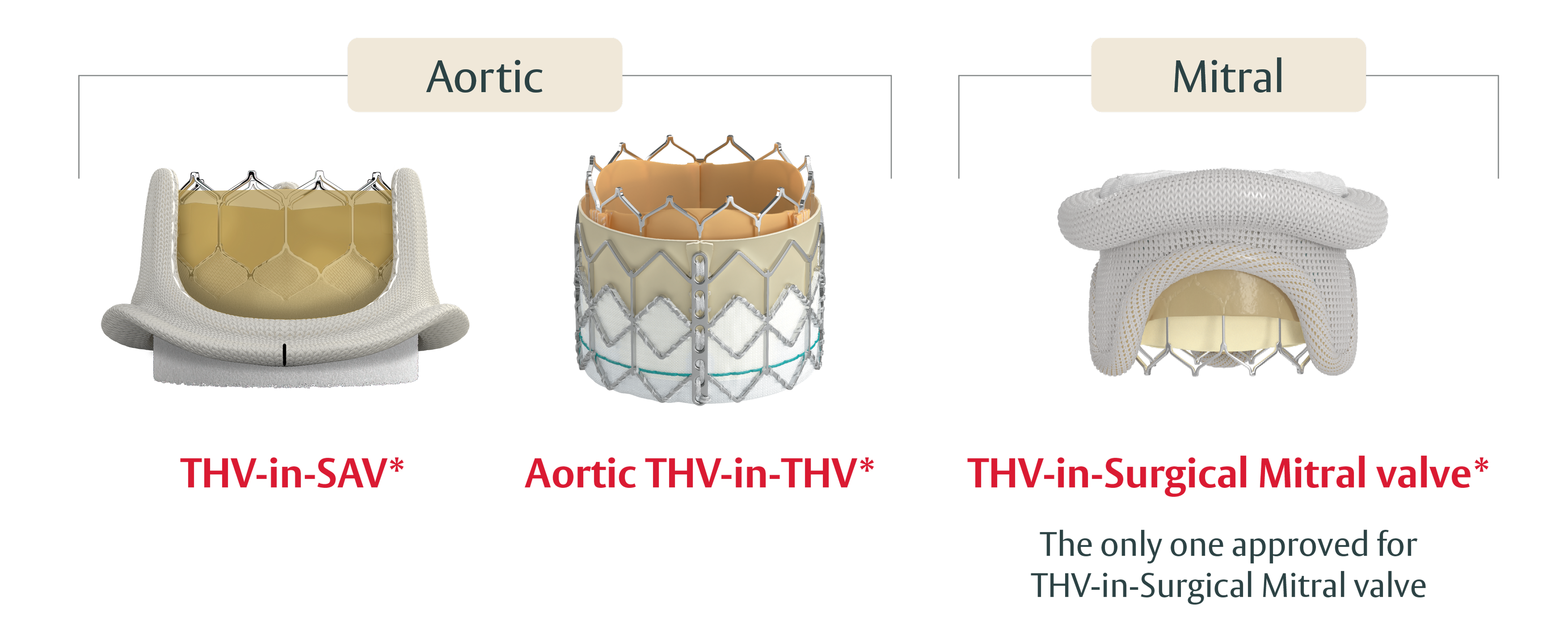
Designed to host future valve reinterventions
100% Coronary access thanks to:10
- Low frame design
- Open cell geometry
*Risk of sinus sequestration if (1) prior TAV commissure level above STJ and (2) the distance between TAV and STJ was <2.0mm in each coronary sinus.
.png)