Edwards Benchmark Program
Learn about the six meaningful clinical outcomes our program targets and how it can deliver quality of life and hospital savings for your center.
Find out more
Learn about the six meaningful clinical outcomes our program targets and how it can deliver quality of life and hospital savings for your center.
Find out more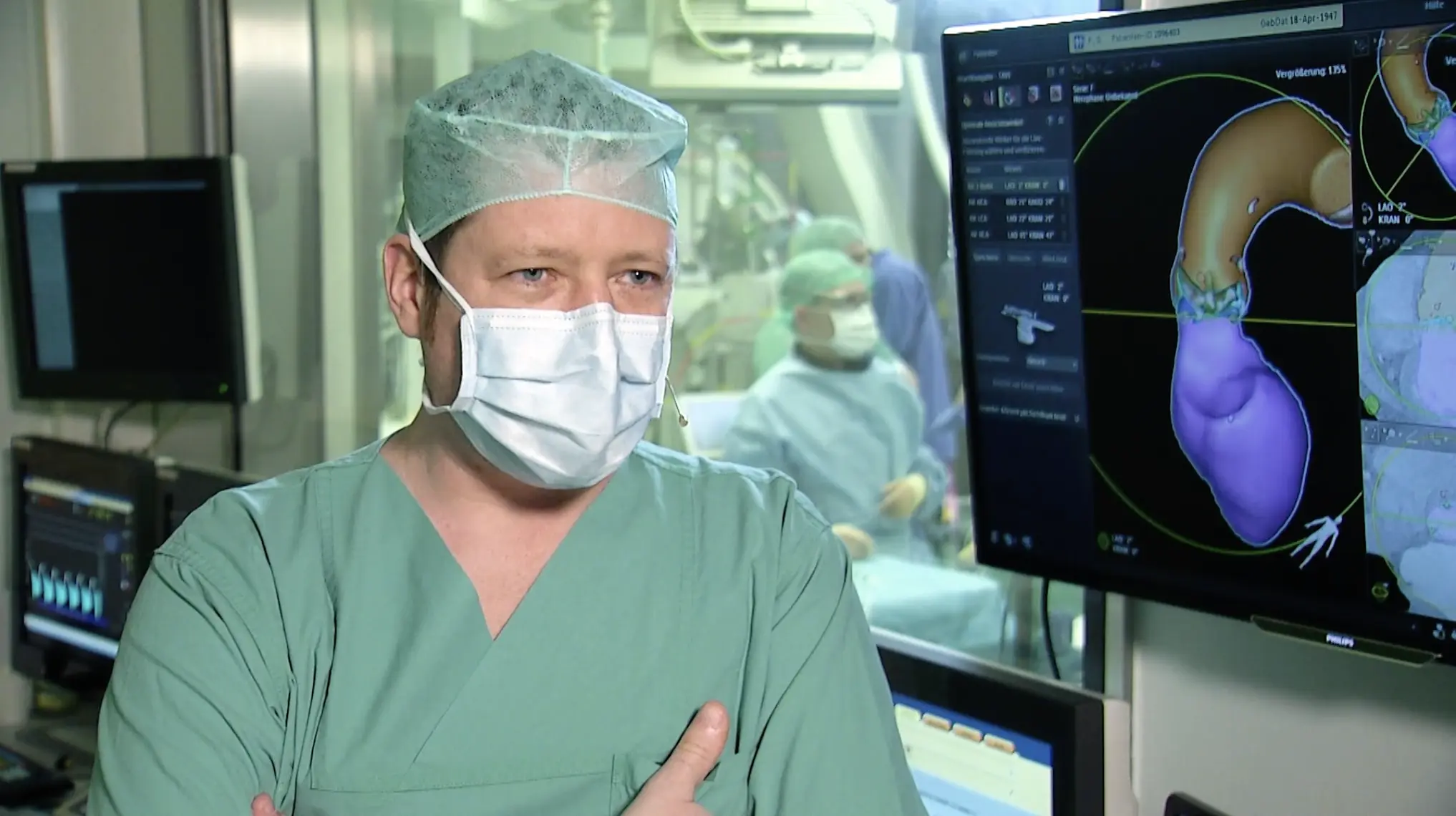
Learn about Edward's options for Bicuspid TAVI with interesting clinical data, recorded sessions and cases, and interviews of influential TAVI leaders.
Find out more
Explore case recordings, expert-delivered educational videos on TAVI procedural challenges, TAVI tips and techniques, peer interviews and more.
View videos
Edwards Learning Network, an innovative and engaging educational portal for healthcare professionals.
About Learning PortalThank you for signing up to the heartvalves.com newsletter. You will now receive emails on the latest developments and industry insights on heart valve innovation and technology.
Medical device for professional use. For a listing of indications, contraindications, precautions, warnings, and potential adverse events, please refer to the Instructions for Use (consult eifu.edwards.com where applicable).
PP--EU-3352 v2.0