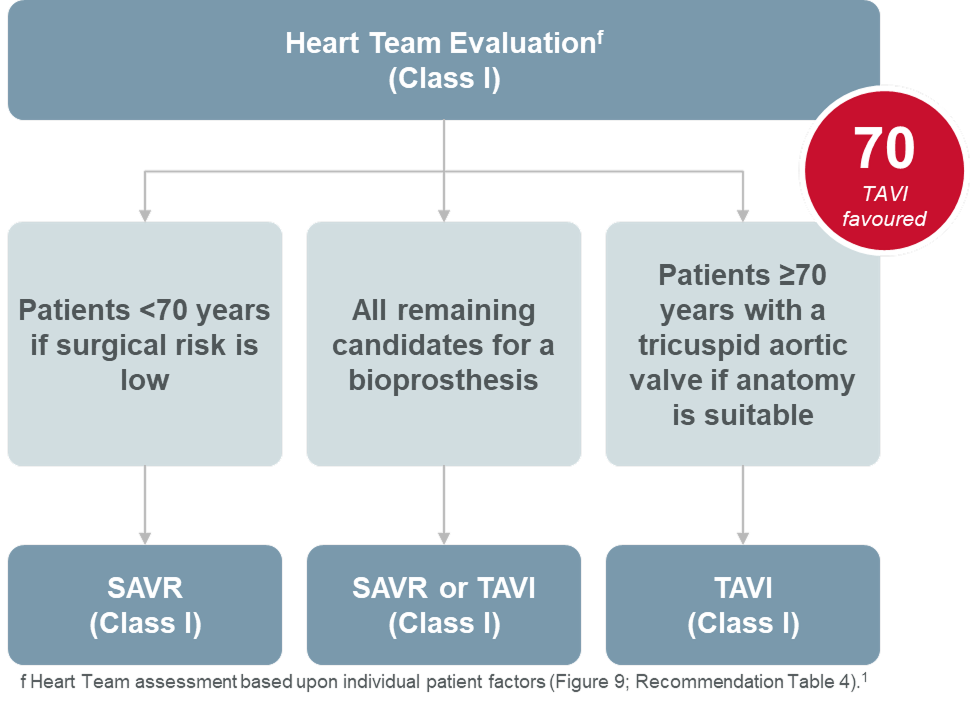
The SAPIEN 3 Ultra RESILIA valve
Advanced calcium-blocking tissue technology5,6
Potential to improve valve longevity and reduce risk of reintervention*
Taller, textured outer skirt extended to 29mm valve5
Delivering the PVL results you demand impacting immediate and long-term outcomes11,12
Only THV with dry tissue storage5,6
Mitigates calcium-attracting glutaraldehyde residuals6
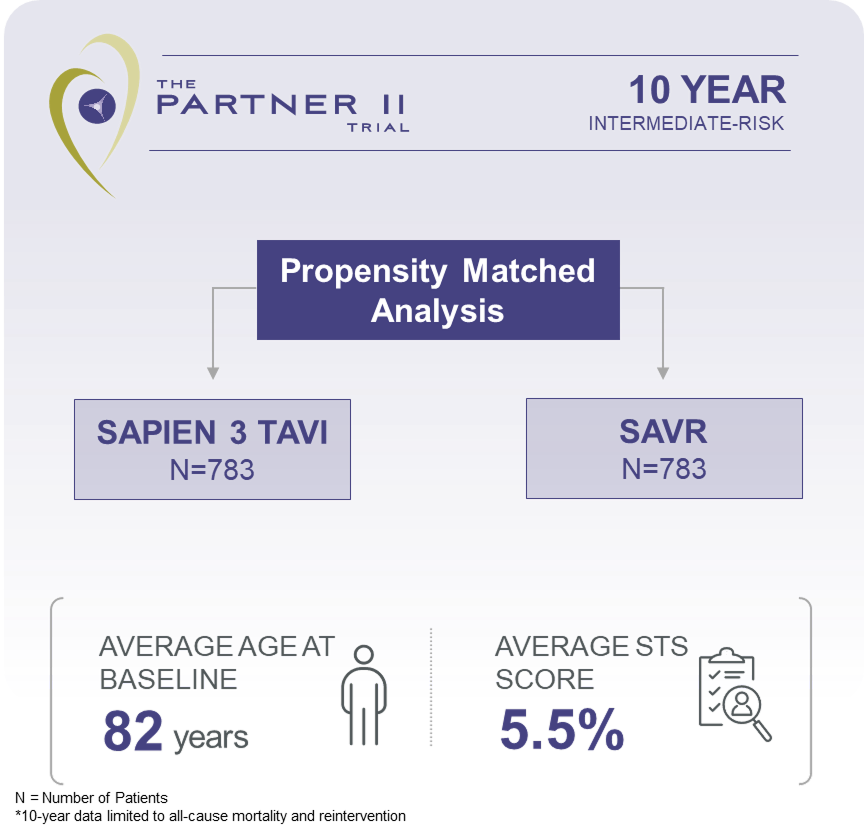
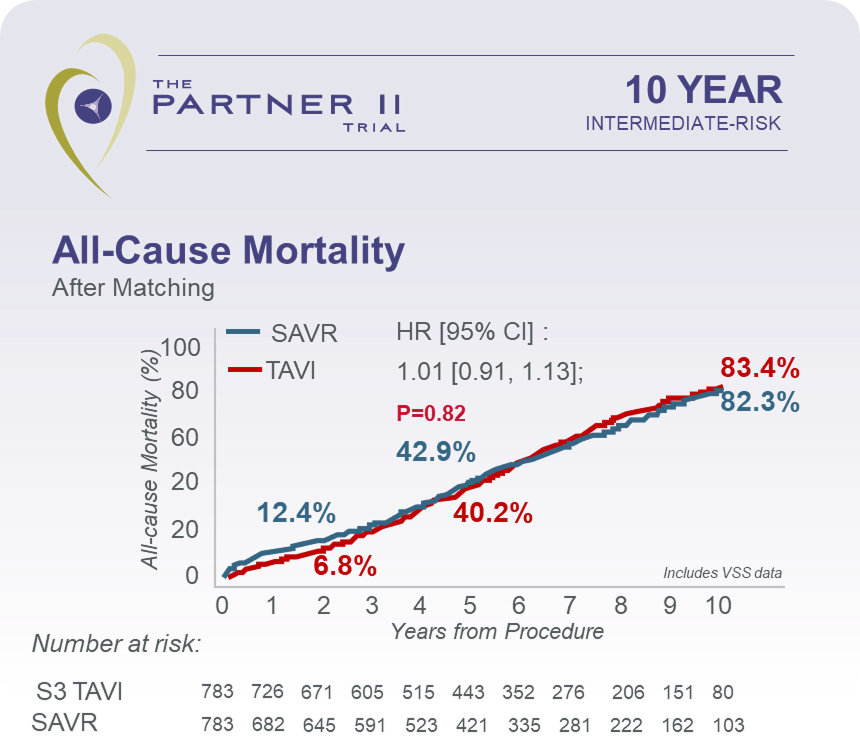
Explore clinical data for RESILIA tissue*No clinical data are available that evaluate the long-term impact of RESILIA tissue in TAVI patients.